Market Strategy
NeuRewards Market Strategy
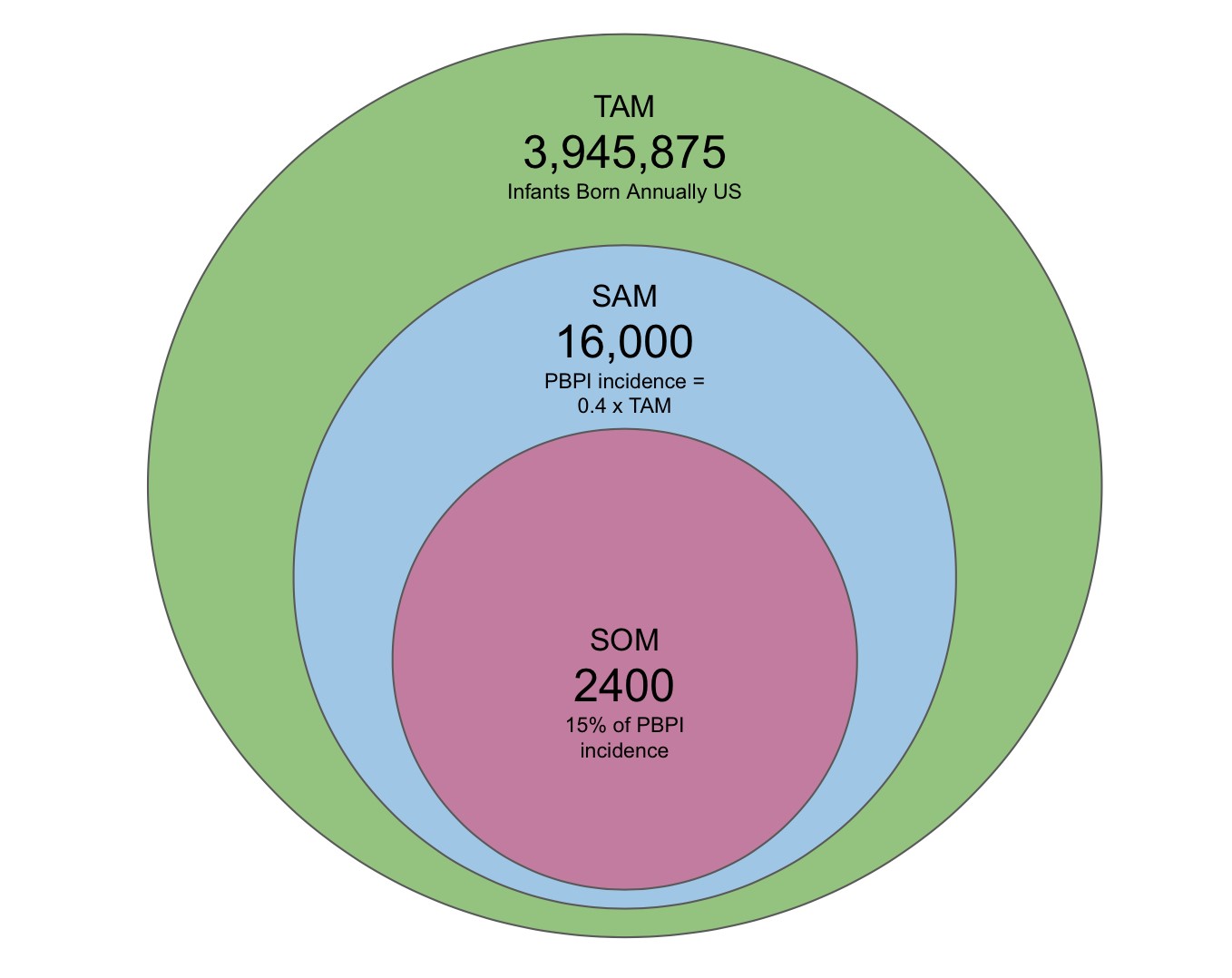
NeuReward’s target market is the parents of infants with PBPI. Once an infant is diagnosed with PBPI, the physical therapist will tell them of our device and parents can buy it in store. Through aggressive marketing by therapist endorsement and NeuRewards hopes to reach 15% of our serviceable available market.
5 Year Plan
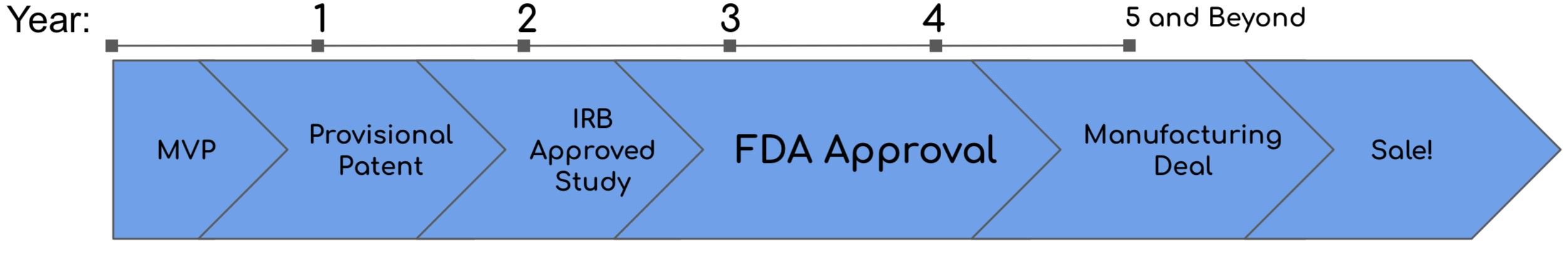
Currently NeuRewards has achieved a minimum viable product (MVP). A provisional patent is in the works to grant the Lodestar Innovations team the time and motivation to further develop NeuRewards. In the upcoming months, NeuRewards will be tested on infants in a research setting after Internal Review Board Approval is achieved. At the end of this trial, it will be determined whether or not FDA approval is needed for such a device. If so, then the rest of the 5 year process will be to gain FDA approval.